TULIP QULISA HCV KIT
TULIP QULISA HCV KIT Specification
- Instruments Type
- IVD (In Vitro Diagnostic) kit
- Usage Type
- Diagnostic Laboratory
- Storage Instructions
- Store at 2C to 8C in a dry environment, keep away from direct sunlight
- Measurement Range
- Qualitative detection
- Shelf Life
- 12 months from manufacturing date
- Function
- Detection of anti-HCV antibodies
- Accuracy
- >99% (as per manufacturers validation)
- Frequency
- Single use per test
- Features
- High specificity and sensitivity, ready-to-use reagents, easy protocol, reliable results
- Equipment Type
- HCV ELISA Test Kit
- Material
- Medical-grade plastic and reagents
- Condition
- New
- Technology
- Enzyme-linked immunosorbent assay (ELISA)
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Detection of Hepatitis C Virus (HCV) antibodies in human serum or plasma
- Dimension (L*W*H)
- Standard Kit Packaging
- Weight
- Approx. 500g - 700g (varies by pack size)
- Color
- White and Blue packaging
- Specificity
- >99%
- Sensitivity
- >99%
- Test Principle
- Based on indirect ELISA for the detection of anti-HCV
- Sample Type
- Serum or Plasma
- Incubation Time
- 60 Minutes (variable as per step)
- Pack Size
- 96 Tests/Kit
- Kit Contents
- Microwell strips, conjugate, wash buffer, substrate, stop solution, positive and negative controls, instruction manual
About TULIP QULISA HCV KIT
Advanced Diagnostic Excellence for Healthcare
TULIP QULISA HCV KIT is expertly designed for use in diagnostic laboratories, providing accurate qualitative detection of anti-HCV antibodies in human serum or plasma. Its primary application lies in the reliable identification of Hepatitis C Virus exposure, making it vital for screening and confirmatory testing. Key features include ready-to-use reagents, robust medical-grade materials, and a rapid 60-minute incubation time. The kit's easy protocol and elite performance suit frequent laboratory use, ensuring seamless integration into diagnostic workflows.
Supply Availability, Certifications, and Delivery Insights
Elevate your laboratory's testing capabilities with steady supply ability and assurance on the TULIP QULISA HCV KIT. Recognized for stringent quality standards and valid certifications, this solution meets global diagnostic requirements. The list price reflects competitive valuation for a product of its class, ensuring excellent value at the asking price. Standard delivery timelines ensure timely order fulfillment and uninterrupted operations, cementing its place among dependable suppliers in India's IVD market.
FAQ's of TULIP QULISA HCV KIT:
Q: How does the TULIP QULISA HCV KIT detect Hepatitis C Virus antibodies?
A: The kit operates on the indirect ELISA principle, identifying anti-HCV antibodies present in human serum or plasma using enzyme-linked detection for top-tier accuracy.Q: What makes this HCV ELISA test kit the preferred choice for diagnostic labs?
A: With over 99% sensitivity and specificity, a straightforward testing protocol, and ready-to-use reagents, the kit ensures reliable, ravishing results while minimizing errors and saving time.Q: When should the kit be used for optimal results?
A: It is optimal for routine screening or confirmation of Hepatitis C Virus infection in clinical and diagnostic laboratory settings, delivering results within approximately 60 minutes.Q: Where should the kit be stored for best preservation?
A: Store the kit in a dry place, maintaining a temperature between 2C to 8C, and keep it away from direct sunlight to ensure reagent stability and prolonged shelf life.Q: What is the process for conducting an HCV test with this kit?
A: Testing involves adding the sample and reagents to microwell strips as instructed, followed by specified incubation and washing steps before interpreting qualitative results.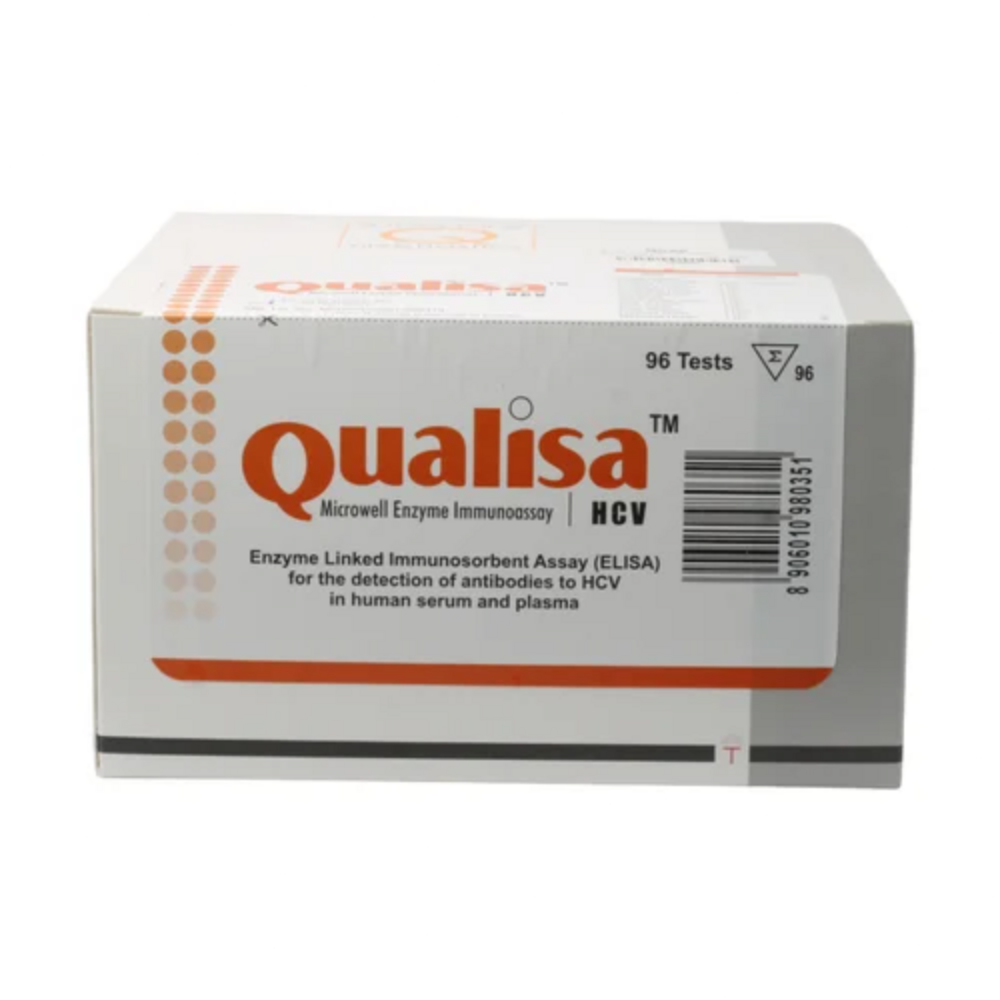

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
DIAMOND MARCURIAL BLOOD PRESSURE APPEARANCE
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Metal and Glass (Mercury Column), Rubber Bulb and Tube, Nylon Cuff
Use : Blood Pressure Measurement
Color : Grey Body, Black Cuff
Weight : Approx. 900 grams
PEEP VALVES HO
Price 1550 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Highquality medicalgrade polymer and stainless steel spring
Use : To maintain positive end expiratory pressure in respiratory circuits
Color : Blue and White
Weight : Approx. 30 grams
CHEST DRAINAGE SET
Price 1950 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Medical Grade PVC
Use : Thoracic Drainage/Chest Drainage Procedures
Color : Transparent with graduated markings
Weight : Lightweight
CENTRAL VENOUS CATHETER
Price 1550 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Polyurethane / Medical Grade Plastic
Use : Venous Access for Drug and Fluid Administration
Color : Clear / Transparent with Colored Identification Hub
Weight : Lightweight (approximately 1030g per catheter)




 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
