SD BIOLINE ROTAVIRUS RAPID TEST CARD
SD BIOLINE ROTAVIRUS RAPID TEST CARD Specification
- Features
- Quick, easy-to-use, visual result interpretation
- Function
- Rapid detection of rotavirus antigen
- Usage Type
- Single-use
- Storage Instructions
- Store at 1-30C, avoid freezing
- Instruments Type
- In vitro diagnostic test
- Display Type
- Visual line display
- Accuracy
- Diagnostics sensitivity >99%, specificity >99%
- Shelf Life
- 18-24 months (unopened)
- Frequency
- Single Test
- Measurement Range
- Qualitative (Positive/Negative)
- Equipment Type
- Diagnostic Rapid Test Card
- Material
- Plastic, test strip membrane
- Condition
- New
- Technology
- Immunochromatographic assay
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Operating Type
- Manual
- Use
- Detection of rotavirus antigen in human fecal specimens
- Dimension (L*W*H)
- Standard card size (approx. 95 mm x 24 mm)
- Weight
- Approx. 10 grams per test card
- Color
- White (card), color bands for result
- Sample Type
- Human fecal specimen
- Result Time
- 510 minutes
- Interpretation
- Two lines = positive, one line = negative, no line or invalid control = invalid
- Lot Number/Expiry Date
- Printed on package
- Packaging
- Individually packed in foil pouches with desiccant
- Storage Humidity
- <80% RH (recommended)
- Test Principle
- Colloidal gold conjugate based immunoassay
- Intended User
- Medical professionals, laboratory personnel
About SD BIOLINE ROTAVIRUS RAPID TEST CARD
SD Bioline Rotavirus Rapid TestThe SD BIOLINE ROTAVIRUS RAPID TEST CARD is an immunochromatographic assay for the detection of Group A rotavirus in fecal specimens. It utilizes two kinds of antibody in a solid phase sandwich immunochromatography to detect group-specific proteins, including the major inner capsid protein, present in Group A rotaviruses.
Key Features:- Rapid test: Results are available within 10-20 minutes
- Immunochromatographic assay
- Detects Group A rotavirus in fecal specimens
- High sensitivity and specificity
- Includes instruction leaflet
- Suitable for use in hospitals and diagnostic labs
- Material: Plastic card
- Specimen: Fecal specimens (about 50mg)
- Result time: 10-20 minutes
- Brand: SD BIOLINE
- Manufacturer: Standard Diagnostics, Inc.
The SD BIOLINE Rota/Adeno Rapid kit has been evaluated in several studies, demonstrating acceptable analytical and clinical performances. One study found that the kit detected different genotypes of rotavirus and showed high agreement rates with molecular testing methods. Another study evaluated the kits performance in detecting rotavirus and adenovirus simultaneously, demonstrating acceptable analytical and clinical performances.
Conclusion:The SD BIOLINE ROTAVIRUS RAPID TEST CARD is a reliable and rapid diagnostic tool for detecting Group A rotavirus in fecal specimens. Its high sensitivity and specificity make it a valuable asset for hospitals and diagnostic labs, enabling timely diagnosis and treatment of rotavirus infections.
Fast and Simple Rotavirus Detection
Designed for speed and ease, the SD BIOLINE ROTAVIRUS RAPID TEST CARD delivers results within 10 minutes. Its straightforward procedure and visual line display mean that minimal training is required for reliable use, making it suitable for a wide range of healthcare settings. Portability and single-use design add to its practicality, supporting efficient diagnosis and patient management.
Accurate and Reliable Outcomes
The test uses immunochromatographic assay technology, delivering greater than 99% sensitivity and specificity. Each test card is precisely manufactured for consistency, with quality assurance through lot number and expiry date labeling. Results are simple to interpret: two lines for positive, one for negative, and invalid if no control line appears.
Convenient Storage and Handling
Each test is packed in a foil pouch with desiccant, requiring storage between 1-30C and humidity below 80% RH. The robust packaging ensures long shelf life (18-24 months, unopened) and maintains test integrity until use. No additional instruments are required, allowing immediate deployment in various environments.
FAQ's of SD BIOLINE ROTAVIRUS RAPID TEST CARD:
Q: How is the SD BIOLINE ROTAVIRUS RAPID TEST CARD used for rotavirus detection?
A: The card is used by applying a processed sample from a human fecal specimen onto the test strip. After waiting 5-10 minutes, the presence of colored lines indicates whether the rotavirus antigen is detected or not, following the interpretation guidelines provided.Q: What are the main benefits of using this rapid test card?
A: The key benefits include swift results (within 10 minutes), easy-to-read visual interpretation, high diagnostic accuracy, and portable single-use packaging. These factors facilitate rapid clinical decisions and infection control.Q: When should this test be used?
A: The SD BIOLINE ROTAVIRUS RAPID TEST CARD is intended for use when rotavirus infection is suspected in a patient, particularly in cases of acute gastroenteritis, as part of routine diagnostic protocols in healthcare settings.Q: Where should the test kits be stored for optimal performance?
A: Store the test cards in a dry environment at temperatures between 1-30C and humidity below 80% RH. Avoid freezing to preserve the test's functionality and shelf life.Q: What is the process for interpreting test results?
A: Observe the result window after 5-10 minutes: two visible colored lines indicate a positive result, one line denotes a negative result, and the absence of control lines signifies an invalid test that should be repeated.Q: Who is the intended user of this rapid test card?
A: This product is designed for use by medical professionals and laboratory personnel experienced in diagnostic testing and interpretation of fecal specimen results.Q: What should be done if a test card fails to show control lines?
A: If no control line appears, the test result is invalid. In such cases, repeat the test with a new card and ensure the procedure and storage instructions are strictly followed.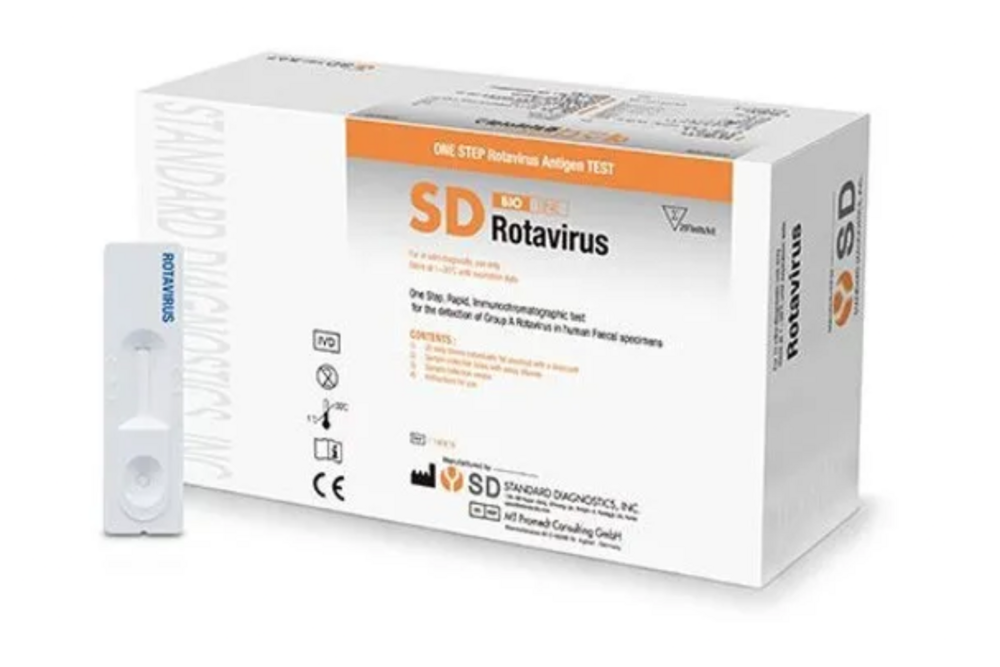

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
Meril Anti A, Anti B, Anti D Blood Grouping Test Kit
Price 500 INR / Piece
Minimum Order Quantity : 10 Pieces
Weight : Approx. 2530 g per vial
Instruments Type : Blood Typing Reagent Kit
Condition : New
Shelf Life : 24 Months from Manufacturing
RESUSCITATION KIT NEONATAL
Price 1550 INR / Piece
Minimum Order Quantity : 10 Pieces
Weight : Approx. 0.8 kg
Instruments Type : Manual resuscitator set
Condition : New
Shelf Life : 5 years (if stored properly)
TONSIL FORCEPS PLAIN TOOTH
Price 1800 INR / Piece
Minimum Order Quantity : 10 Pieces
Weight : Approx. 80 grams
Instruments Type : Forceps
Condition : New
Shelf Life : Indefinite with Proper Care
Fluoroplastic Offset Total Prosthesis ( TORP )
Price 1450 INR / Piece
Minimum Order Quantity : 10 Pieces
Weight : Extremely Lightweight (few milligrams)
Instruments Type : Surgical Prosthesis
Condition : New
Shelf Life : 5 Years (unopened sterile packaging)





 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
