MEDTRONIC LAUCHER GUIDING CATHETER
MEDTRONIC LAUCHER GUIDING CATHETER Specification
- Usage Type
- Hospital, Clinical
- Accuracy
- High positioning precision
- Features
- Large inner lumen, excellent torque control, soft tip, radiopaque marker
- Instruments Type
- Medical Catheter
- Storage Instructions
- Store in a dry, cool place, away from direct sunlight
- Function
- Provides a stable pathway for interventional devices
- Shelf Life
- Up to 3 years
- Equipment Type
- Guiding Catheter
- Material
- Polyamide (Nylon)
- Condition
- Sterile, Single-use
- Technology
- Catheterization, Vascular Access
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual
- Use
- Cardiology, endovascular procedures
- Dimension (L*W*H)
- Varies by model (typically 100 cm length)
- Weight
- Lightweight, < 10 g
- Color
- Transparent with colored coded hubs
- Tip Shapes
- Multiple available (Judkins Left/Right, Amplatz, etc.)
- Hub Type
- Color coded for French size identification
- Available Sizes (French)
- 5F, 6F, 7F, 8F
- Radiopacity
- Distal marker for visualization under fluoroscopy
- Sterilization Method
- Ethylene Oxide (EtO)
- Pressure Rating
- Up to 1200 psi maximum
About MEDTRONIC LAUCHER GUIDING CATHETER
Launcher coronary guide catheters come with the performance advantages you expect from a market leader including a wide variety of curve shapes for transradial and femoral percutaneous coronary intervention (PCI).
Additional Information:
- Item Code: LA6EBU35
- Production Capacity: 100
- Delivery Time: 1 DAY DELHI NCR
- Packaging Details: BOX
Versatility for Diverse Procedures
The Launcher Guiding Catheter supports a range of tip shapes and French sizes, allowing adaptability in various cardiology and endovascular procedures. Its compatibility ensures healthcare professionals can select the ideal configuration for each intervention.
Enhanced Visualization and Safety
The catheter is equipped with a distal radiopaque marker, providing real-time visualization under fluoroscopy. This, combined with its precise positioning and soft tip, optimizes safety and accuracy during device manipulation.
User-Friendly and Reliable Design
Featuring color-coded hubs for quick size identification, the catheter's lightweight polyamide construction ensures smooth handling. Its single-use, sterile packaging and three-year shelf life make it a dependable option for busy clinical settings.
FAQ's of MEDTRONIC LAUCHER GUIDING CATHETER:
Q: How are the different Medtronic Launcher Guiding Catheter sizes identified?
A: Each catheter features a color-coded hub system that corresponds to its French size, enabling quick and easy identification during procedures.Q: What tip shapes are available for the Launcher Guiding Catheter?
A: Tip shapes include Judkins Left, Judkins Right, Amplatz, and others, offering flexibility for various vascular anatomies and procedures.Q: When should the Launcher Guiding Catheter be used in clinical settings?
A: This catheter is intended for use during cardiology or endovascular interventions, providing a stable pathway for interventional devices in hospital or clinical environments.Q: Where should the catheter be stored before use?
A: Store the catheter in a dry, cool place away from direct sunlight to maintain product integrity and sterility.Q: What is the process for visualizing the catheter during a procedure?
A: A radiopaque distal marker integrated into the soft tip enhances visibility under fluoroscopy, allowing for precise placement and navigation in real time.Q: What are the main benefits of using the Launcher Guiding Catheter?
A: Key benefits include high positioning precision, large inner lumen for easy device passage, silent operation, and superior torque control, ensuring effective and safe interventions.Q: How is the Launcher Guiding Catheter sterilized and is it reusable?
A: The catheter is sterilized using the Ethylene Oxide (EtO) method and is intended for single-use only to minimize infection risks.




Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
DIAMOND MARCURIAL BLOOD PRESSURE APPEARANCE
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Features : Mercury Column, EasytoRead Scale, Accurate Measurement
Wall Mounted : Other, No (Portable Desktop Model)
Weight : Approx. 900 grams
Dimension (L*W*H) : Approx. 330 mm x 110 mm x 50 mm
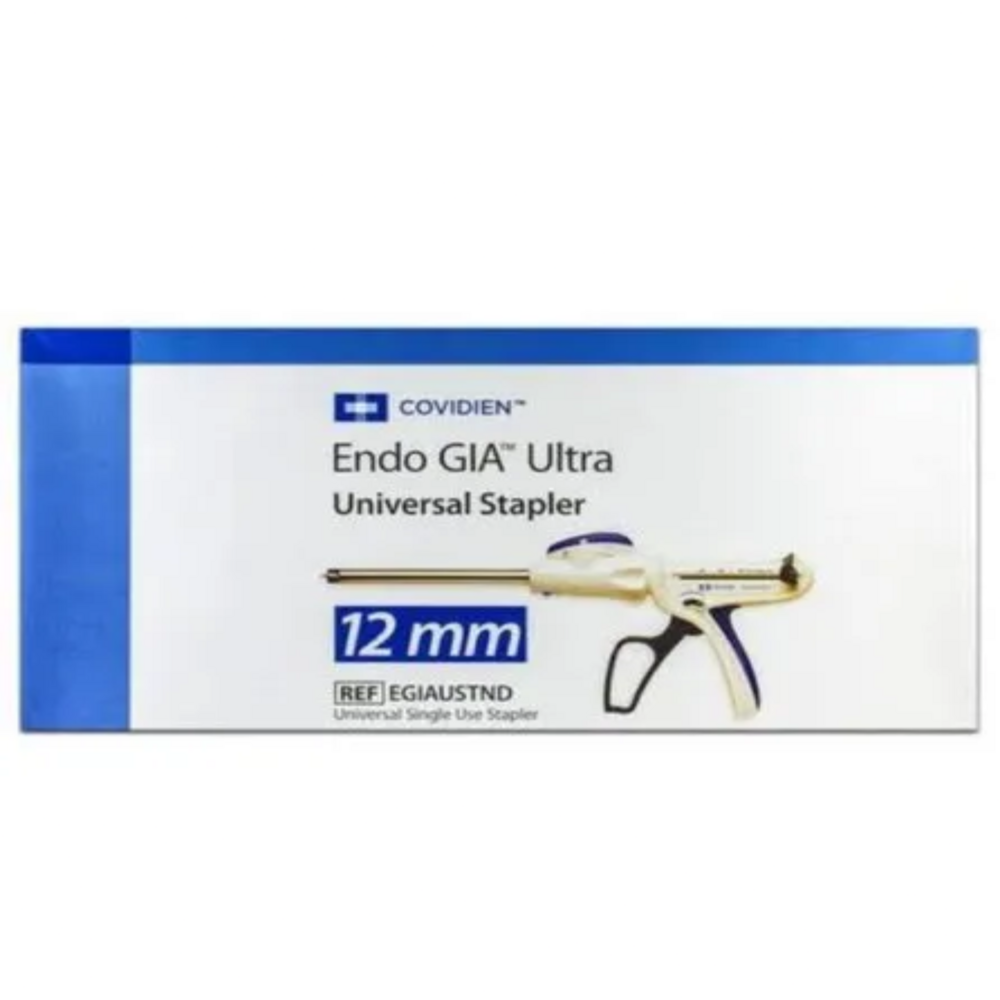
COVIDIEN ENDO GIA ALTTRA UNIVERSAL STAPLER
Price 1650 INR / Piece
Minimum Order Quantity : 10 Pieces
Features : Universal handle compatibility, Articulating shaft, Multiple reload lengths, Consistent staple formation, Precise tissue compression
Wall Mounted : No
Weight : Lightweight, Approx. 0.5 kg
Dimension (L*W*H) : Universal 45mm shaft; Cartridge length varies by reload (30mm, 45mm, 60mm)
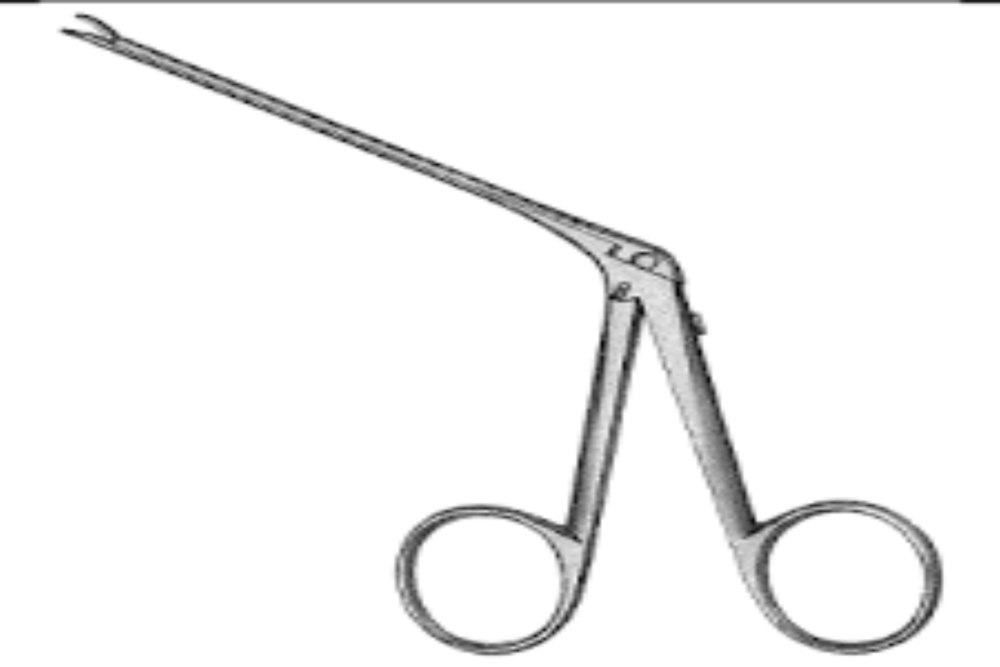
Stapes Prosthesis Handling Forcep
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Features : Fine Tips for Precision Grip, Smooth Surface, Antislip Handle, Corrosion Resistant
Wall Mounted : No
Weight : Approx. 20 g
Dimension (L*W*H) : Approximately 12 cm Length
ADISON FORCEPS PLAIN TEETH
Price 1450 INR / Piece
Minimum Order Quantity : 10 Pieces
Features : Precision tip, Light weight, Corrosion resistant
Wall Mounted : No
Weight : Approx. 3040 grams
Dimension (L*W*H) : Approximately 15 cm length




 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
