JMITRA HCV Elisa Test Kit
JMITRA HCV Elisa Test Kit Specification
- Features
- High sensitivity, high specificity, user friendly protocol, rapid processing
- Storage Instructions
- Store at 2C to 8C, avoid freezing
- Instruments Type
- IVD (In-vitro Diagnostic)
- Function
- Detection of Hepatitis C Virus antibodies
- Shelf Life
- 12-24 Months (at 2C to 8C)
- Accuracy
- >99%
- Measurement Range
- Qualitative
- Usage Type
- Laboratory/Diagnostic
- Equipment Type
- HCV Elisa Test Kit
- Material
- Plastic, Paper (Consumables)
- Condition
- New
- Technology
- Enzyme Linked Immunosorbent Assay (ELISA)
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Noise Level
- Silent Operation
- Operating Type
- Manual
- Use
- Detection of HCV antibodies in human serum or plasma
- Power Source
- Not Required (External Reader Required)
- Color
- White/Blue
- Kit Components
- Microwell plate, sample diluent, conjugate, substrate, stop solution, positive/negative controls, wash buffer
- Specificity
- >99.5%
- Test Principle
- Indirect ELISA
- Number of Tests per Kit
- 96 tests per kit
- Sample Type
- Human serum or plasma
- Incubation Temperature
- Room temperature (20C30C)
- Packaging Type
- Box
- Sensitivity
- >99.5%
- Test Time
- Approx. 120 minutes
- Read-out
- Requires ELISA plate reader
About JMITRA HCV Elisa Test Kit
- 3rd Generation Assay
- Unique combination of HCV antigens for E1, E2, Core, NS3, NS4 & NS5
- Break-away microwell strips
- Based on Indirect Elisa principle
- Colour coded reagents to monitor procedural steps.
- Short assay procedure: 90mins
- Longer shelf life: 15 months at 2-8C.
- Sensitivity 100% & Specificity 97.4% by WHO.
Accurate and Reliable HCV Detection
With accuracy exceeding 99% and high specificity, the JMITRA HCV Elisa Test Kit ensures confidence in diagnosing Hepatitis C Virus infection. Its robust performance is validated for laboratory and diagnostic use, adhering to IVD standards for trustworthy results.
Convenient and Comprehensive Kit Components
This kit provides all required consumables, including microwell plates, reagents, and controls. Packaged in a sturdy box, it simplifies inventory management and ensures testers have everything needed for efficient laboratory workflow.
Simple Protocol and Rapid Turnaround
The user-friendly protocol enables completion of testing in approximately 120 minutes at room temperature (20C-30C). Its straightforward procedures support quick training for laboratory personnel and facilitate high throughput in busy diagnostic settings.
FAQ's of JMITRA HCV Elisa Test Kit:
Q: How is the JMITRA HCV Elisa Test Kit used for detecting HCV antibodies?
A: The kit uses an Indirect ELISA technique to detect HCV antibodies in human serum or plasma samples. The process involves adding the sample to microwell plates, followed by incubation and the addition of reagents. A color change read by an ELISA plate reader indicates the presence of antibodies.Q: What sample types are compatible with this HCV Elisa Test Kit?
A: The test is validated for use with human serum or plasma samples, ensuring suitability for most standard clinical and diagnostic laboratory applications.Q: When should the kit be stored, and at what temperature?
A: The JMITRA HCV Elisa Test Kit should be stored between 2C and 8C upon receipt. Avoid freezing the components to maintain performance and reagent integrity.Q: Where is the test intended to be performed?
A: This test is designed for use in laboratory or diagnostic settings equipped with an ELISA plate reader, as it requires controlled environments and professional handling.Q: What is the process duration, and how many samples can be tested per kit?
A: Each test run takes approximately 120 minutes from sample addition to result. A single kit can process up to 96 individual tests.Q: What benefits does the kit provide to laboratories and healthcare facilities?
A: The kit offers rapid, reliable, and highly accurate detection of HCV antibodies, making it ideal for diagnostics. It is packaged with all necessary components, has a long shelf life, and is portable for flexible laboratory usage.Q: How does the kit ensure accuracy and minimize false readings?
A: The kit's high sensitivity and specificity (both >99.5%), along with included positive and negative controls, ensure consistent, accurate results while minimizing the likelihood of false positives or negatives.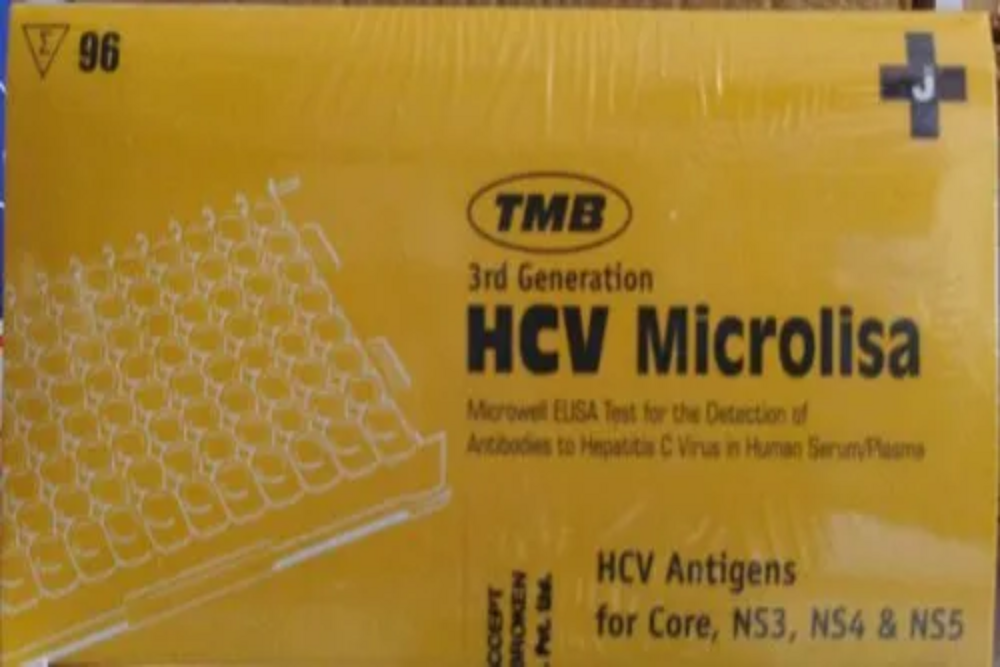

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
INTELIFLO SYSTEMS CIRCUIT
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, HighQuality PCB, Electronic Components
Technology : MicrocontrollerBased System
Weight : Approx. 350 g
Usage Type : Industrial
Niko Electrosurgical Neutral Electrode Cautery Plate301
Price 1450 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Stainless Steel Plate with Polymeric Insulation
Technology : Monopolar Electrosurgery Compatible
Weight : Approx. 270g
Usage Type : Hospital Use, Operation Theatre
HUDSON TYPE CANNULA WITH BONET
Price 1950 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, Medical Grade PVC
Technology : Conventional Moulding
Weight : Lightweight
Usage Type : Single Use, Disposable
CALBIOTECH VITAMIN D ELISA TEST KIT
Price 13000 INR / Piece
Minimum Order Quantity : 10 Pieces
Material : Other, High quality plastic and reagents
Technology : EnzymeLinked Immunosorbent Assay (ELISA)
Weight : Approx. 0.5 to 1 kg depending on kit size
Usage Type : Diagnostic Laboratory Use




 Send Inquiry
Send Inquiry Send SMS
Send SMS
