MERIL DENGUE NS1 ELISA KIT
MERIL DENGUE NS1 ELISA KIT Specification
- Instruments Type
- Immunodiagnostic Test Kit
- Measurement Range
- Qualitative (positive/negative)
- Features
- High sensitivity and specificity, Ready-to-use reagents, Rapid and reliable results, Suitable for mass screening
- Storage Instructions
- Store at 2C to 8C
- Usage Type
- Laboratory Diagnostic Use only
- Shelf Life
- 12 months from the date of manufacture
- Function
- Detection of NS1 antigen in suspected dengue cases
- Accuracy
- >98%
- Equipment Type
- Dengue NS1 ELISA Kit
- Material
- Diagnostic Reagents/Consumables
- Condition
- New
- Technology
- ELISA (Enzyme Linked Immunosorbent Assay)
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- No
- Operating Type
- Manual
- Use
- In-vitro qualitative detection of Dengue NS1 antigen in human serum or plasma
- Power Source
- Not Required (Used in conjunction with standard ELISA reader)
- Dimension (L*W*H)
- Standard ELISA Plate Size (96-well format)
- Weight
- Approx. 0.5 kg (kit)
- Color
- Assorted (as per component reagents)
- Specificity
- >98%
- Test Principle
- Solid-phase ELISA based on capture of NS1 antigen
- Intended Users
- Medical laboratories, Hospitals, Diagnostic centers
- Number of Tests per Kit
- 96 tests
- Result Time
- ~90 minutes
- Regulatory Status
- IVD certified
- Packing Type
- Boxed kit with all consumables
- Compatibility
- Compatible with any standard ELISA reader
- Manufactured By
- Meril Diagnostics Pvt. Ltd.
- Controls Included
- Positive control and Negative control
- Sensitivity
- >97%
- Sample Volume Required
- 100 µl per well
About MERIL DENGUE NS1 ELISA KIT
Merilisa Dengue NS1 Ag is a solid phase enzyme linked immunosorbent assay for the detection of Dengue NS1 antigen in human serum or plasma. The kit detects all four sub types; DEN1, DEN2, DEN3 and DEN4 of Dengue virus.Consistent Accuracy and Reliability
With sensitivity exceeding 97% and specificity above 98%, the MERIL DENGUE NS1 ELISA KIT delivers highly reliable results for early detection of dengue infection. Laboratories benefit from its robust design, IVD certification, and built-in quality controls, ensuring every test performed meets professional standards.
Rapid and Easy Workflow
The kit features ready-to-use reagents and a user-friendly manual operation, requiring only 100 l of sample per well. Complete results are obtained in about 90 minutes, expediting patient diagnosis. Its compatibility with standard ELISA plate readers allows seamless integration into existing laboratory setups.
Comprehensive Kit for Hassle-Free Testing
Each boxed kit comes equipped with all essential consumables, positive and negative controls, and standardized reagents. The kit supports 96 tests and remains stable for 12 months when stored at 2C to 8C, making it efficient and cost-effective for large-scale testing in diverse clinical environments.
FAQ's of MERIL DENGUE NS1 ELISA KIT:
Q: How does the MERIL DENGUE NS1 ELISA KIT detect dengue infections?
A: This kit utilizes a solid-phase ELISA technique to capture and qualitatively detect the NS1 antigen present in human serum or plasma samples. The presence of the NS1 antigen indicates acute dengue infection.Q: What is the sample volume required for each test and what samples can be used?
A: Each test requires 100 l of human serum or plasma. Ensure that samples are properly collected and prepared according to laboratory protocols for accurate results.Q: When can the test results be obtained after starting the assay?
A: The MERIL DENGUE NS1 ELISA KIT provides clear results in approximately 90 minutes, making it suitable for both routine and urgent diagnostic needs.Q: Where can this kit be used and who are the intended users?
A: This kit is intended for use in medical laboratories, hospitals, and diagnostic centers by trained laboratory personnel for in-vitro diagnostic applications.Q: What is the process for running a test using this kit?
A: Simply add the sample to the designated well, follow the manual protocol with ready-to-use reagents, and use any standard ELISA reader to interpret the results. Positive and negative controls are included for quality assurance.Q: What are the key benefits of choosing this kit for dengue screening?
A: Key advantages include high sensitivity and specificity, rapid turnaround time, ready-to-use reagents, and suitability for mass screening-making it ideal for outbreak response and high-throughput environments.Q: How should the kit be stored and what is its shelf life?
A: Store the kit at 2C to 8C. It has a shelf life of 12 months from the manufacturing date, ensuring reliability over extended storage periods.



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
RESECTOSCOPIC TURP SET
Price 1950 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Use : Transurethral Resection of Prostate
Dimension (L*W*H) : 24F & 26F Sheath Sizes (approximate dimensions vary)
Color : Silver with Insulated Handle (Blue/White)
MONOPOLAR ELECTRODES
Price 1950 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Use : Electrosurgical Procedures
Dimension (L*W*H) : Varies by model; Common Length: 1520 cm
Color : Metallic with colored insulation sleeve (blue/black)
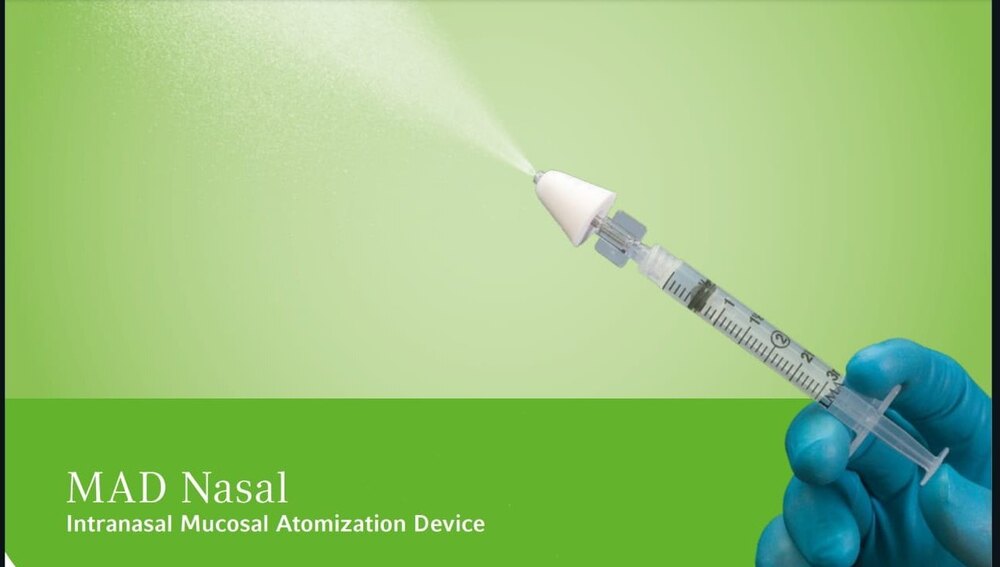
Mad Nasal Intranasal Mucosal Atomization Device
Price 1560 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Use : Intranasal Drug Delivery
Dimension (L*W*H) : Approx. 78 x 33 x 17 mm
Color : Transparent/White
Rebreathing Bag SILICONE
Price 2450 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Use : For assisted breathing in anesthesia and resuscitation
Dimension (L*W*H) : Variable (0.5L, 1L, 2L, 3L sizes available)
Color : Translucent Blue/Green




 Send Inquiry
Send Inquiry Send SMS
Send SMS
