E7509 VALLEYLAB PATIENT PLAT
E7509 VALLEYLAB PATIENT PLAT Specification
- Instruments Type
- Electrosurgical Accessory
- Usage Type
- Single Use/Disposable
- Function
- Provides Safe Return Path of Electrical Current During Surgery
- Features
- Pre-wired, Self-Adhesive, Pre-Gelled, Disposable, Designed for Compatible Electrosurgical Generators
- Frequency
- Compatible with Standard Electrosurgical Unit Frequencies
- Shelf Life
- Up to 3 Years (Unopened)
- Storage Instructions
- Store in a Cool, Dry Place Away from Direct Sunlight
- Equipment Type
- Patient Return Electrode (Patient Plate)
- Material
- Flexible Nonwoven Material with Conductive Gel
- Condition
- New
- Technology
- Monopolar, Disposable
- Portable
- Yes
- Wall Mounted
- No
- Operating Type
- Electrosurgical Accessory
- Use
- For Safe Patient Electrosurgery, Providing Return Path for High-Frequency Currents
- Dimension (L*W*H)
- 21.7 cm x 15.2 cm (approx.)
- Weight
- Lightweight, <80 g
- Color
- Blue/White
- Biocompatibility
- Latex-Free, Hypoallergenic
- Compatibility
- Compatible with Valleylab and Most Major ESU Brands
- Disposal
- Biohazard Waste, Single Patient Use
- Patient Age Group
- Adult
- Sterility
- Yes, Sterile Packaged
- Attachment Method
- Self-Adhering/Adhesive
- Packaging
- Individually Packed
- Model Number
- E7509
About E7509 VALLEYLAB PATIENT PLAT
Valleylab patient return electrodes are recognized worldwide for quality, safety, and innovative technology. All return electrodes are manufactured with our patented PolyHesive II high moisture, conductive adhesive hydrogel that provides the performance of a gel without the gel mess.
PolyHesive II hydrogel benefits- Water-based hydrogel lowers skin resistance.
- Unique elongation fills patient skin irregularities and minimizes contact voids to improve the total electrical conduction area.
- Surface area disperses current over large area, minimizing heat buildup.
- Acrylic adhesive improves pad contact quality and provides a moisture barrier to significantly reduce the potential for fluid invasion.
- Adult, infant (<30 lb.), and neonatal (<6 lb.) sizes available.
- Non-irritating* and non-sensitizing*
- The exclusive Valleylab REM system monitors patient impedance levels, automatically deactivating the generator if a fault at the patient return electrode is detected.
- REM is an integral part of the Valleylab Full Circuit system standardization program.
Additional Information:
- Item Code: E7509
- Production Capacity: 500pc
- Delivery Time: 1 day delhi ncr
- Packaging Details: box
Reliable Electrosurgical Safety Solution
Engineered for compatibility with major ESU generators, the E7509 ensures a safe and effective return path for electrosurgical currents. Its flexible, hypoallergenic design and pre-gelled conductive layer provide secure patient contact, while the sterile, disposable packaging supports infection control protocols.
Sterile and Hypoallergenic Patient Friendly Design
Manufactured from latex-free, nonwoven materials, the E7509 accommodates sensitive skin and reduces the risk of allergic reactions. Each electrode is individually packaged to maintain sterility, making it suitable for single-use procedures in adult patients.
FAQ's of E7509 VALLEYLAB PATIENT PLAT:
Q: How is the E7509 VALLEYLAB PATIENT PLAT used during surgery?
A: The E7509 is applied to the patient's skin before monopolar electrosurgical procedures to provide a safe return path for high-frequency currents. Its adhesive backing ensures secure placement, and it should remain in place for the duration of the surgery.Q: What are the storage requirements for this patient plate?
A: It should be stored in a cool, dry place away from direct sunlight to maintain sterility and effectiveness. Unopened, the E7509 has a shelf life of up to three years.Q: When should the E7509 be disposed of, and how?
A: Dispose of the E7509 after a single use, or if its sterile packaging is compromised. It must be placed in biohazard waste according to hospital safety protocols.Q: Where can the E7509 be used, and with what equipment is it compatible?
A: This electrode is designed for use in clinical or surgical environments and is compatible with Valleylab and most major electrosurgical unit (ESU) brands.Q: What are the benefits of using a hypoallergenic, latex-free patient plate?
A: A hypoallergenic and latex-free design minimizes the risk of allergic reactions, making the E7509 suitable for a wide range of patients, including those with latex sensitivities.Q: How does the self-adhesive, pre-gelled design improve the application process?
A: The pre-gelled, self-adhesive backing simplifies placement, ensures better skin contact, and helps achieve reliable electrical conductivity during the surgical procedure.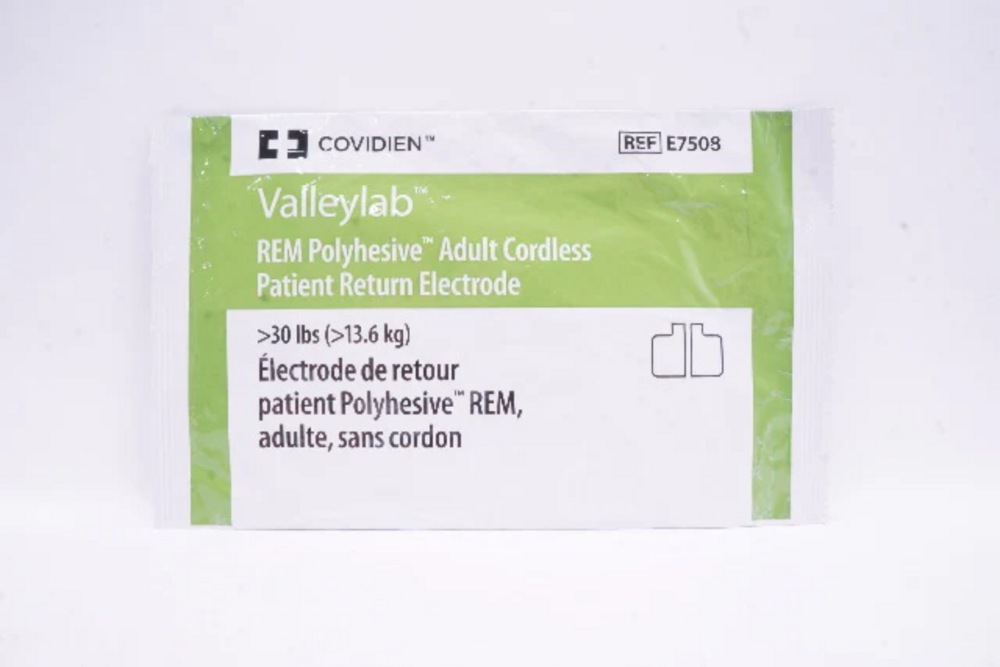

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
Percutaneous Dilation Tracheostomy Kit with blue
Price 14500 INR / Piece
Minimum Order Quantity : 10 Pieces
Shelf Life : Up to 5 years (unopened, from manufacturing date)
Storage Instructions : Store in a cool, dry, sterile environment
Use : Tracheostomy procedure (bedside and ICU)
Portable : Yes
Adult Blockbuster E T Tube
Price 1050 INR / Piece
Minimum Order Quantity : 10 Pieces
Shelf Life : 5 years
Storage Instructions : Store in a cool, dry place, away from direct sunlight
Use : Airway Management
Portable : Yes
UTERINE FORCEPS
Price 2500 INR / Piece
Minimum Order Quantity : 10 Pieces
Shelf Life : Long Shelf Life (with proper care)
Storage Instructions : Clean and sterilize after each use, store in dry place
Use : Gynecological Surgical Procedures
Portable : Yes
ADHESIVE REMOVER SPRAY
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Shelf Life : 24 Months
Storage Instructions : Store In Cool, Dry Place
Use : Removes Sticky Residue and Adhesives
Portable : Yes




 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
