DIASYS HOMOCYSTEINE FS
DIASYS HOMOCYSTEINE FS Specification
- Instruments Type
- Clinical Chemistry Analyzers
- Measurement Range
- 150 mol/L (varies by analyzer)
- Shelf Life
- 12 months (at 2-8C)
- Function
- Quantitative determination of homocysteine
- Accuracy
- High, with good correlation to reference methods
- Storage Instructions
- Store at 28C. Protect from light.
- Usage Type
- Laboratory diagnostic
- Features
- High precision, liquid-stable, ready-to-use, compatible with most clinical chemistry analyzers
- Equipment Type
- Diagnostic Reagent Kit
- Material
- Liquid-stable reagent chemicals
- Condition
- New
- Technology
- Enzymatic Cycling Method
- Portable
- Yes (as a kit for laboratory use)
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Operating Type
- Automated or Manual on compatible analyzers
- Use
- In vitro determination of total homocysteine in human serum or plasma
- Color
- Colourless to pale yellow solutions
- Sample Type
- Serum or EDTA/heparin plasma
- Intended User
- Professional/laboratory personnel
- Calibrator/Control Required
- Yes, specific for homocysteine
- Assay Time
- ~10 minutes per test (on automated systems)
- CE Marking
- Yes
- Principle
- Two reagent, enzymatic cycling assay with photometric detection at 340 nm
- Safety Information
- Not for internal or therapeutic use
- Kit Presentation
- 2 x 25 mL, 2 x 12.5 mL, and other sizes available
- Packaging
- Sealed bottles in protective carton
About DIASYS HOMOCYSTEINE FS
Homocysteine FS is an enzymatic test to determine homocysteine in serum and plasma.The amino-acid homocysteine represents an independent risk factor for cardiovascular diseases (CVD).
Rapid and Accurate Homocysteine Testing
The DIASYS HOMOCYSTEINE FS kit provides high-precision results for determining total homocysteine levels in serum or plasma samples. With an assay time of approximately 10 minutes on automated systems, laboratories can deliver quick and reliable results. The technology is compatible with a wide array of clinical chemistry analyzers, ensuring versatility across various laboratory settings.
Simple Workflow and Flexible Kit Presentations
Featuring liquid-stable, ready-to-use reagents, this kit streamlines daily laboratory processes. Calibrators and controls specific for homocysteine are required, further enhancing result reliability. DIASYS HOMOCYSTEINE FS is available in multiple pack sizes (such as 2 x 25 mL or 2 x 12.5 mL), enabling laboratories to choose the optimal volume for their workload.
Robust, Safe, and Certified for Laboratory Use
Packaged in sealed bottles within protective cartons, the kit maintains stability for up to 12 months when stored at 2-8C and protected from light. It holds CE marking, ensuring conformity to European standards, and is strictly for in vitro diagnostic use by professional personnel. The reagents are colorless to pale yellow, user-friendly, and compatible with both automated and manual operation.
FAQ's of DIASYS HOMOCYSTEINE FS:
Q: How is the DIASYS HOMOCYSTEINE FS assay performed?
A: The assay uses an enzymatic cycling method involving two liquid-stable reagents. After adding serum or EDTA/heparin plasma to the reagents, the reaction occurs and is measured photometrically at 340 nm. The process takes about 10 minutes per test on compatible automated chemistry analyzers.Q: What sample types are compatible with this kit?
A: The DIASYS HOMOCYSTEINE FS kit is intended for use with human serum or plasma samples collected with EDTA or heparin as anticoagulants.Q: When should calibrators and controls be used in the assay?
A: Specific calibrators and controls for homocysteine must be used with every assay run to ensure accuracy and validity of the results, as required for clinical diagnostics.Q: Where should I store the DIASYS HOMOCYSTEINE FS reagents?
A: Reagents must be kept at 2-8C and protected from light to maintain their shelf life of up to 12 months. They should remain in their sealed bottles until usage.Q: What are the key benefits of using this kit in the laboratory?
A: The kit offers high accuracy, rapid turnaround, ready-to-use reagents, and compatibility with a broad range of chemistry analyzers. Its robust packaging and liquid stability reduce waste and streamline workflow for laboratory personnel.Q: Can DIASYS HOMOCYSTEINE FS be used manually as well as on automated analyzers?
A: Yes, the kit is suitable for both manual operation and use on compatible automated clinical chemistry analyzers, offering flexibility for different laboratory setups.



Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
Curved I VIEW VIDEO LARENGOSCOP
Price 12500 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Material : Other, Stainless Steel Blade, Medical Grade Plastic Handle
Use : Intubation, Airway Examination
Color : White & Blue
DIAMOND MARCURIAL BLOOD PRESSURE APPEARANCE
Price 1250 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Material : Other, Metal and Glass (Mercury Column), Rubber Bulb and Tube, Nylon Cuff
Use : Blood Pressure Measurement
Color : Grey Body, Black Cuff
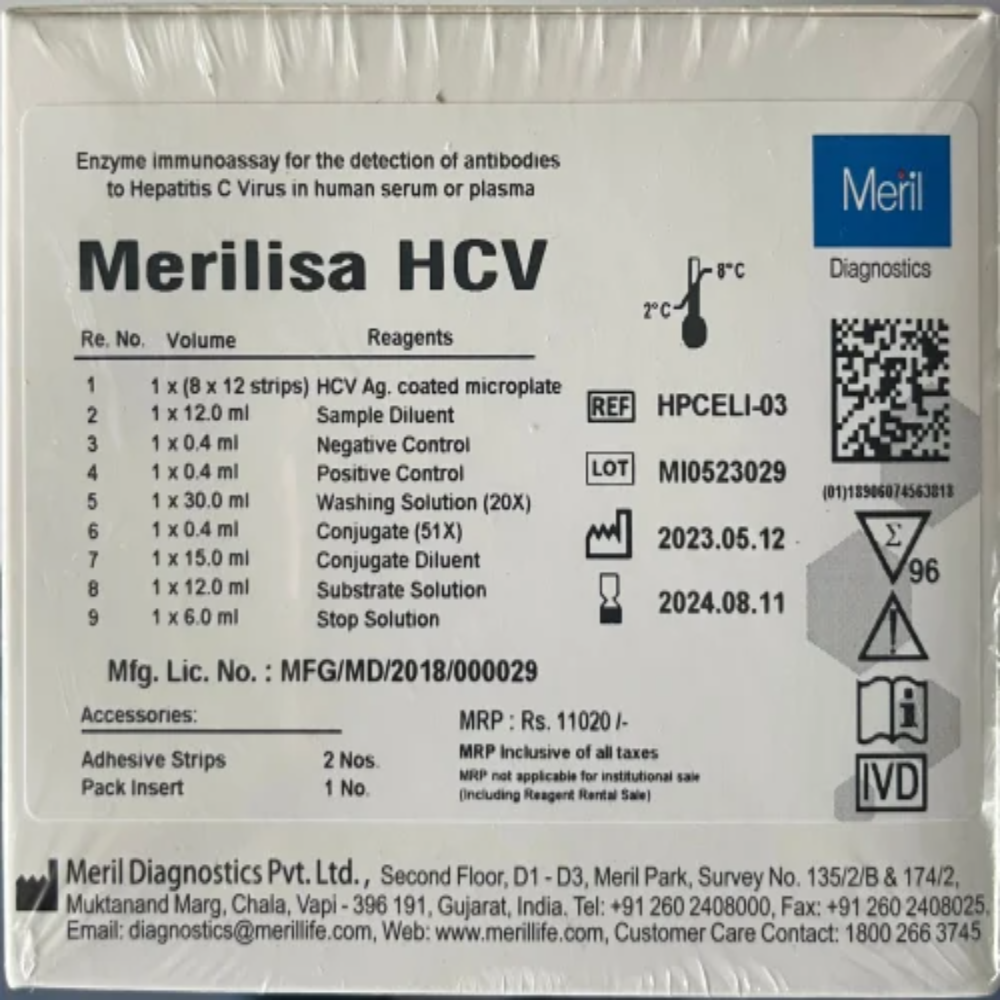
MERIL HCV ELISA TEST KIT
Price 2000 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Material : Other, Plastic, highquality reagents, microtiter plate
Use : Qualitative detection of antibodies to Hepatitis C Virus (antiHCV) in human serum or plasma
Color : White (Kit components: colorless/reagent colored as per protocol)
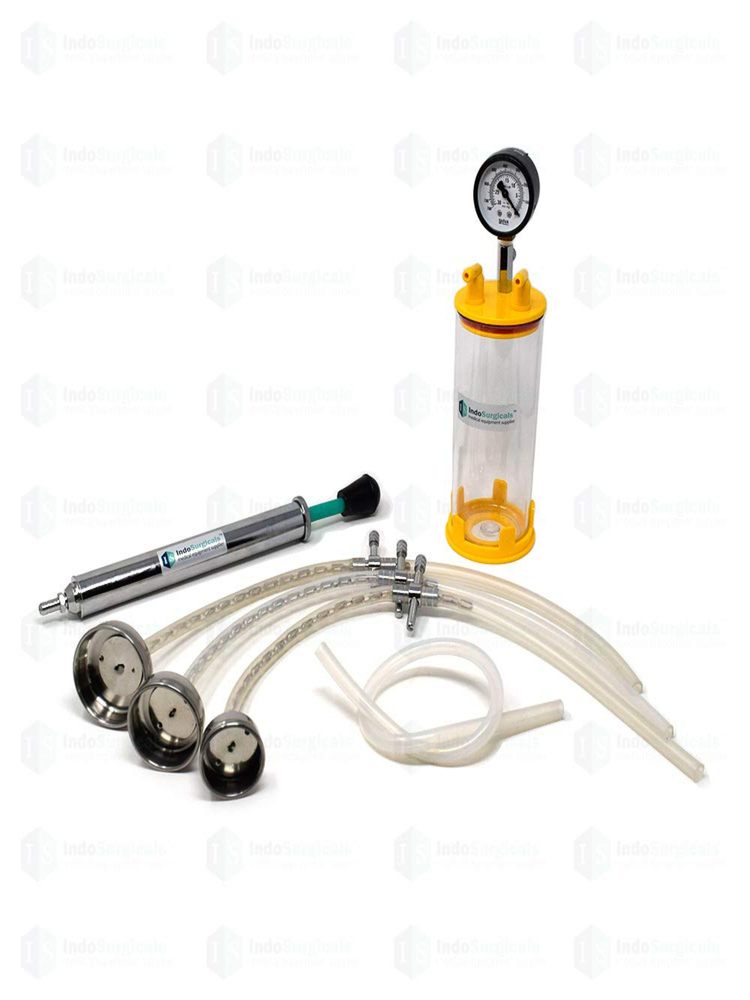
VACUMM EXTRACTOR (MAIMSTROM TYPE)
Price 2600 INR / Piece
Minimum Order Quantity : 10 Pieces
Condition : New
Material : Other, Stainless Steel & Medical Grade Components
Use : Obstetrical Procedures, Assisted Vaginal Delivery
Color : Metallic Grey




 Send Inquiry
Send Inquiry Send SMS
Send SMS
