3M TEGADERM 1633IN
3M TEGADERM 1633IN Specification
- Shelf Life
- 3 Years
- Function
- Wound Cover and IV Site Fixation
- Storage Instructions
- Store in Cool, Dry Place, Away from Direct Sunlight
- Usage Type
- Single Use
- Features
- Breathable, Waterproof, Bacteria and Virus Barrier, Hypoallergenic, Conformable
- Instruments Type
- Medical Dressing
- Equipment Type
- Transparent Film Dressing
- Material
- Polyurethane Film
- Condition
- New
- Technology
- Adhesive Film Technology
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent
- Operating Type
- Manual Application
- Use
- Wound Protection and IV Site Coverage
- Dimension (L*W*H)
- 10 cm x 12 cm
- Weight
- Lightweight
- Color
- Transparent
- Sterility
- Sterile EO
- Model
- Tegaderm 1633IN
- Latex Content
- Latex-Free
- Permeability
- Moisture Vapor Permeable
- Application Area
- IV Catheter Sites, Surgical Incisions, Minor Burns, Abrasions
- Quantity per Box
- 50 Pieces
- Sterilization Method
- Ethylene Oxide Gas
- Product Code
- 1633IN
- Adhesive Type
- Acrylic Adhesive
About 3M TEGADERM 1633IN
3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for patients with implanted venous ports. 3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for use with implanted venous ports and non-coring Huber needlesAdditional Information:
- Item Code: 1633
- Delivery Time: 1 Day
- Packaging Details: 100Pcs in the Box
- 3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for patients with implanted venous ports. 3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for use with implanted venous ports and non-coring Huber needles
Additional Information:
- Item Code: 1633
- Delivery Time: 1 Day
- Packaging Details: 100Pcs in the Box
- 3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for patients with implanted venous ports. 3M Tegaderm I.V. Transparent Film Dressing with Adhesive-Free Window is designed for use with implanted venous ports and non-coring Huber needles
Additional Information:
- Item Code: 1633
- Delivery Time: 1 Day
- Packaging Details: 100Pcs in the Box
Advanced Wound and IV Protection
Experience secure and reliable wound coverage with Tegaderm 1633IN. Its transparent, breathable, and waterproof properties ensure optimal healing conditions while protecting from bacteria and viruses. The dressing's gentle, acrylic adhesive provides comfortable adherence without irritating the skin.
Versatile Application Areas
This dressing is ideal for a variety of uses, including IV catheter protection, surgical incisions, minor burns, and abrasions. Its conformable polyurethane film adapts easily to the body, maintaining flexibility during patient movement and simplifying routine wound monitoring.
Sterile and Hypoallergenic Solution
Produced under strict sterilization using Ethylene Oxide Gas, Tegaderm 1633IN is latex-free and hypoallergenic, reducing allergic reactions and ensuring patient safety. Each piece is individually packed for single use, guaranteeing sterility until ready for application.
FAQ's of 3M TEGADERM 1633IN:
Q: How should the 3M TEGADERM 1633IN dressing be applied?
A: Before application, ensure the skin is clean and dry. Remove the dressing from its sterile packaging, peel off the protective lining, and gently place the adhesive side over the intended area, such as an IV catheter site or wound. Smooth down the edges to secure it in place.Q: What are the main benefits of using Tegaderm 1633IN for wound or IV site protection?
A: This dressing offers key benefits such as transparency for visual inspection, a waterproof and bacteria/virus-proof barrier, breathability for moisture balance, hypoallergenic properties, and a lightweight, conformable design that promotes patient comfort and healing.Q: When should I use Tegaderm 1633IN film dressing?
A: Tegaderm 1633IN is ideal for use on IV catheter sites, surgical incisions, minor burns, and abrasions. It is recommended whenever a sterile, moisture-vapor permeable, transparent dressing is required for effective wound or IV site coverage.Q: Where can this product be effectively utilized in the medical setting?
A: The dressing is suitable for hospitals, clinics, and home healthcare environments, particularly for covering intravenous insertions, surgical wounds, and superficial skin injuries. It supports both inpatient and outpatient care.Q: What is the sterilization process for Tegaderm 1633IN?
A: Each Tegaderm 1633IN dressing is sterilized using Ethylene Oxide Gas (EO), ensuring that the product remains free from contaminants and safe for direct application to wounds or IV sites.Q: Is Tegaderm 1633IN safe for individuals with sensitive skin or latex allergies?
A: Yes, Tegaderm 1633IN is latex-free and features a hypoallergenic acrylic adhesive, making it suitable for individuals with sensitive skin and those allergic to latex.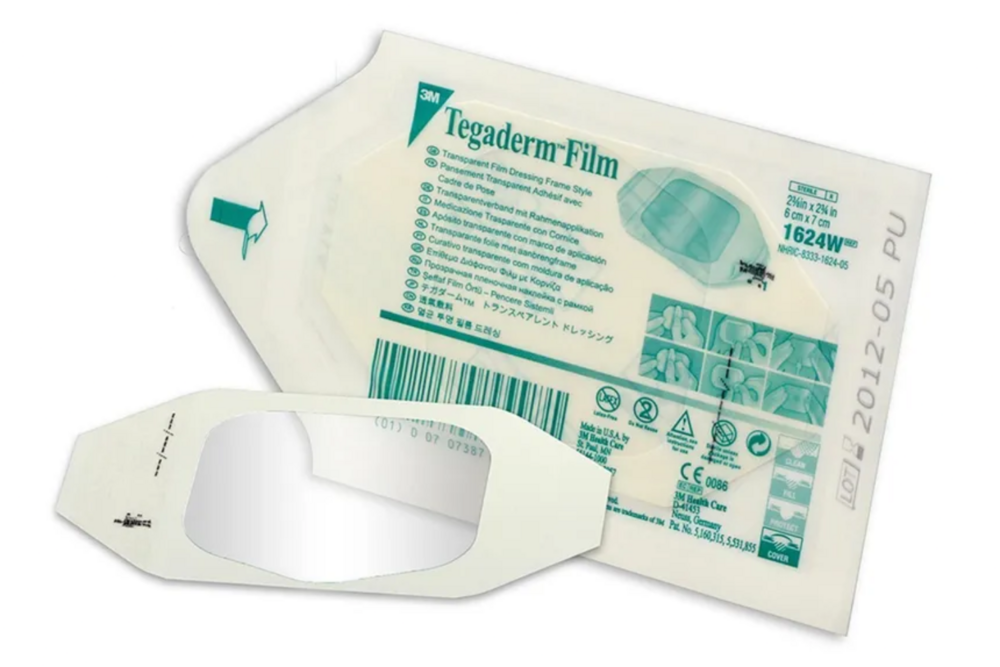

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
Internal Bi-Valve Splints
Price 2000 INR / Piece
Minimum Order Quantity : 10 Pieces
Real-Time Operation : Yes
Instruments Type : Orthopaedic splint
Features : Bivalve design, radiotranslucent, easy application and removal
Storage Instructions : Store in a cool, dry place
BPC FLOWMETER RANGE
Price 1450 INR / Piece
Minimum Order Quantity : 10 Pieces
Real-Time Operation : Yes
Instruments Type : Medical Flowmeter
Features : Clear Scale, Anti Splash, Smooth Flow, Precise Adjustability
Storage Instructions : Store in clean dry place
silicone bubble cpap mask
Price 1450 INR / Piece
Minimum Order Quantity : 10 Pieces
Real-Time Operation : Yes
Instruments Type : Respiratory Care
Features : Soft Cushioning, LeakProof Seal, Adjustable Headgear
Storage Instructions : Store in Cool, Dry Place
JAMSHIDHI BONE MARROW BIOPSY NEEDLE 11GX10CM
Price 1950 INR / Piece
Minimum Order Quantity : 10 Pieces
Real-Time Operation : Yes
Instruments Type : Medical/Surgical Instrument
Features : Sharp tip for easy penetration, Ergonomic handle for comfort, Adjustable depth guard, Luer lock for syringe attachment
Storage Instructions : Store in a cool, dry place away from sunlight




 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free
