I MASK AIRWAY
I MASK AIRWAY Specification
- Function
- Maintains patient airway during anesthesia, resuscitation, and emergency care
- Instruments Type
- Airway management device
- Storage Instructions
- Store in a cool, dry place, away from direct sunlight
- Usage Type
- Single-use
- Shelf Life
- 5 years from date of manufacture
- Frequency
- Single-use
- Accuracy
- High conformity to patient airway
- Measurement Range
- Sizes 1 to 5
- Features
- Soft-seal cuff, flexible airway tube, anatomical shape
- Equipment Type
- I-MASK AIRWAY (Laryngeal Mask Airway)
- Material
- Medical-grade silicone
- Condition
- New
- Technology
- Single-use, non-inflatable cuff design
- Portable
- Yes
- Wall Mounted
- No
- Real-Time Operation
- Yes
- Noise Level
- Silent operation
- Operating Type
- Manual
- Use
- Airway management during anesthesia and emergency
- Power Source
- Not required (manual)
- Dimension (L*W*H)
- Varies by size (Size 1-5 available)
- Weight
- Lightweight, varies by size
- Color
- Transparent or milky white
- Connector Size
- 15 mm standard
- Reusability
- Disposable (single use only)
- Sterility
- Sterile, ready to use
- Intended Patient Age
- Pediatric and adult sizes available
- Radiopacity
- X-ray detectable line included
- Packaging
- Individually packed in sterile pouch
- Manufacturer Detail
- CE & ISO certified
- Insertion Depth Markings
- Provided for accurate placement
- Latex Content
- Latex-free
- Cuff Type
- Non-inflatable, soft-seal cuff
About I MASK AIRWAY
Product Size Suitable for Product Code
1.00 2-5 kg MSI-1318
1.50 5-10 kg MSI-1318
2.00 10-20 kg MSI-1318
2.50 20-30 kg MSI-1318
3.00 30-50 kg MSI-1318
4.00 50-70 kg MSI-1318
5.00 70-100 kg MSI-1318
Tube design conforms to the Nasopharyngeal anatomy.
Made from Non Toxic
Single Use & Disposable Packing : Blister Pape
Reliable Airway Management Solution
The I MASK AIRWAY is engineered for both pediatric and adult patients, providing secure airway management during surgical anesthesia and emergency interventions. Its non-inflatable, soft-seal cuff ensures a high level of conformity to the patient's airway, reducing risks of leakage or displacement.
Single-Use Sterility and Safety
Each I MASK AIRWAY is supplied individually packed in a sterile pouch, ensuring safety from cross-contamination. Its disposable design, along with CE and ISO certifications, upholds the highest standards of patient care and infection control.
User-Friendly and Accurate Placement
This airway device features insertion depth markings and a standard 15 mm connector for compatibility with anesthesia breathing circuits. The inclusion of an X-ray detectable line aids verification of placement, optimizing clinical outcomes across a range of scenarios.
FAQ's of I MASK AIRWAY:
Q: How is the I MASK AIRWAY used during anesthesia or emergencies?
A: The I MASK AIRWAY is manually inserted through the mouth to maintain an open airway in patients undergoing anesthesia, resuscitation, or emergency care. Its anatomical shape and soft-seal, non-inflatable cuff ensure a secure fit and effective ventilation.Q: What patient sizes does the I MASK AIRWAY accommodate?
A: This device is available in a full range of sizes (1 to 5) for both pediatric and adult patients, providing clinicians flexibility to match the airway device to individual anatomical requirements.Q: When should the I MASK AIRWAY be used instead of other airway devices?
A: The I MASK AIRWAY is ideal for situations requiring quick, reliable, and less invasive airway management, particularly during anesthesia induction, short surgical procedures, or when endotracheal intubation is challenging or contraindicated.Q: Where should the I MASK AIRWAY be stored before use?
A: It should be stored in a cool, dry place away from direct sunlight. This preserves the sterility and functionality of the device throughout its 5-year shelf life.Q: What process ensures the sterility and safety of the I MASK AIRWAY?
A: Each unit is individually packed in a sterile pouch and manufactured under CE and ISO certification, ensuring safety, sterility, and adherence to international quality standards.Q: Can the I MASK AIRWAY be reused?
A: No, this laryngeal mask airway is intended for single use only and must be disposed of after one procedure to prevent cross-contamination and uphold patient safety.Q: What are the benefits of the non-inflatable cuff design?
A: The non-inflatable, soft-seal cuff conforms naturally to the airway without the need for inflation, simplifying use, minimizing pressure-related complications, and enhancing patient comfort during airway management.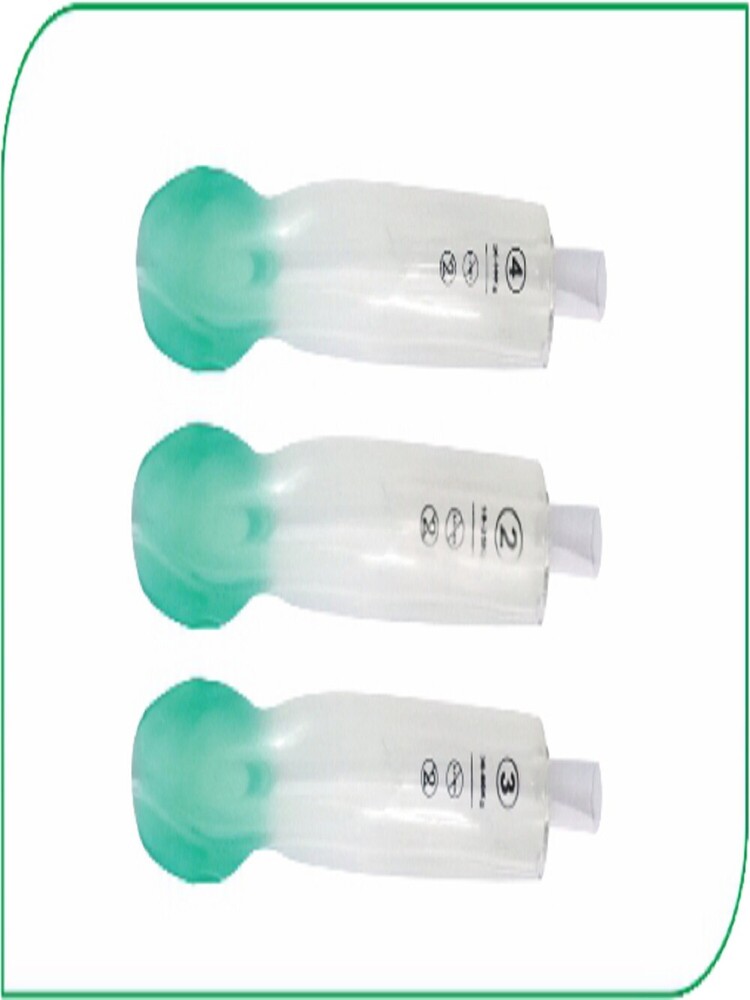

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Intubation Devices And Accessories Category
ENTEROCHECK WB TYPHOID RAPID TEST CARD
Price 2000 INR / Piece
Minimum Order Quantity : 10 Pieces
Use : Detection of Typhoid IgG/IgM antibodies in human whole blood/serum/plasma
Usage Type : Single Use, Diagnostic
Storage Instructions : Store at 2C 30C in a dry place, away from direct sunlight
Material : Other, Plastic, nitrocellulose membrane, immunological reagents
NEO LEAD
Price 1650 INR / Piece
Minimum Order Quantity : 10 Pieces
Use : Cardiac Monitoring, Diagnostic ECG Recording
Usage Type : Single Use
Storage Instructions : Store in a cool, dry place
Material : Other, Foam/Nonwoven backing, Ag/AgCl Sensor
TULIP ERYBANK ANTI-H LECTIN
Price 500 INR / Piece
Minimum Order Quantity : 10 Pieces
Use : Blood Grouping; Detection of H antigen on red blood cells
Usage Type : Laboratory Diagnostics
Storage Instructions : Store at 2C 8C. Do not freeze. Protect from light.
Material : Other, Liquid (Biological reagent)
ADULT MAPLESON CIRCUITS
Price 1550 INR / Piece
Minimum Order Quantity : 10 Pieces
Use : Anaesthesia, Respiratory Care
Usage Type : Hospital, Clinic, Critical Care
Storage Instructions : Store in Cool, Dry Place Away from Direct Sunlight
Material : Other, Medical Grade PVC, Silicone





 Send Inquiry
Send Inquiry Send SMS
Send SMS
